Overview
In 2025, India reinforced its position as a central contributor to the Codex Alimentarius Commission. The Food Safety and Standards Authority of India (FSSAI) led the development of standards across multiple committees, focusing on technical data and scientific evidence. The year demonstrated India's continued commitment to setting global benchmarks that protect public health while supporting fair trade practices. India's national positions, as illustrated in Fig 1, were developed through a structured consultation process. The respective Codex Coordination Groups coordinated inputs from scientists, industry, and consumer groups to reach consensus. By using information on local dietary patterns and preferences and farming conditions, India ensured that the proposed standards were scientifically valid and practical for implementation in diverse food systems.
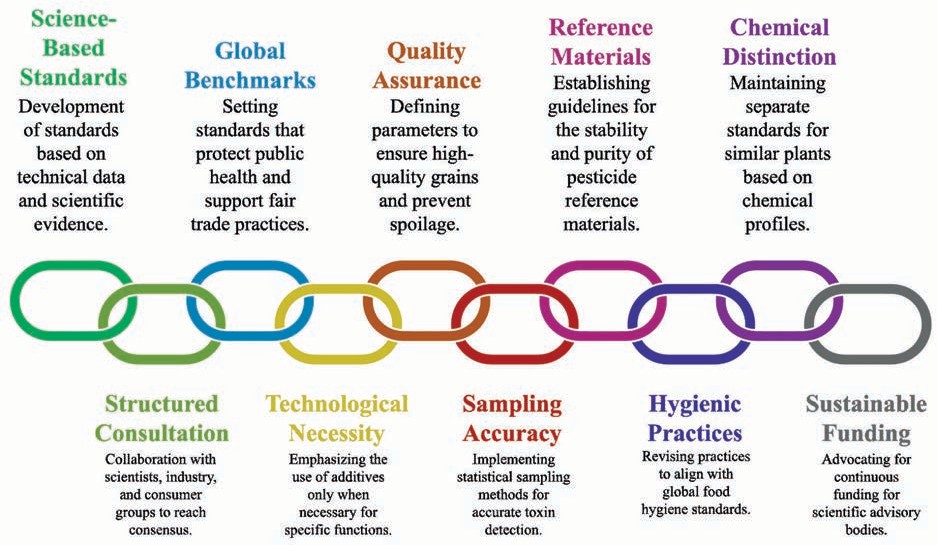
Fig 1: India in Codex in 2025
Fresh Dates (Step 8), Fresh Curry Leaves (Step 5/8), Co-Chair: Fresh Turmeric & Fresh Broccoli
90+ technical comments, "technological necessity" principle for food additives
Chaired EWG for whole millet grains group standard
Chaired EWG on sampling plans for aflatoxins & ochratoxin A in spices (Step 8), Acrylamide Code of Practice
Guidelines for Pesticide Reference Materials (Step 8), CXL for tebuconazole in cumin (0.9 mg/kg)
Chaired EWG on Regional Code of Hygienic Practice for Street-Vended Foods in Asia
Standards for dried coriander seeds, vanilla, large cardamom; opposed merging cassia & cinnamon
Re-elected to Executive Committee (Asia), adoption of Fresh Dates standard & Pesticide Reference Materials guidelines
Codex Committee on Fresh Fruits and Vegetables (CCFFV23) – Mexico City
At the 23rd session of the Codex Committee on Fresh Fruits and Vegetables (CCFFV23) in Mexico City, India led the standard for Fresh Dates to final adoption at CAC48 at Step 8, and the standard for Fresh Curry Leaves to Step 5/8. These standards set technical parameters – such as moisture limits and maturity indices – to prevent spoilage during transport. India also accepted the Co-Chair role for new work on Fresh Turmeric and Fresh Broccoli.
Codex Committee on Food Additives (CCFA55) – Seoul
At the 55th session of the Codex Committee on Food Additives (CCFA55) in Seoul, India emphasized the principle of "technological necessity." The delegation argued that additives should only be used if they serve a specific function, even if safety limits (ADIs) allow for higher amounts. Submitting over 90 technical comments, India supported minimizing chemical use to the lowest level required, reducing the overall chemical load on consumers.
Codex Committee on Cereals, Pulses and Legumes (CCCPL11)
As a major producer of millets, India chaired the EWG for the Codex group standard for whole millet grains during the 11th session of the Codex Committee on Cereals, Pulses and Legumes (CCCPL11). This work addresses quality factors like protein and crude fibre. The developing standard defines moisture and safety parameters to differentiate high-quality grains, which is essential for the global trade of this crop.
Codex Committee on Contaminants in Foods (CCCF18) – Bangkok
In Bangkok, at the 18th session of the Codex Committee on Contaminants in Foods (CCCF18), the focus was on testing methods. Because toxins in spices often occur in "hotspots" (uneven distribution), India chaired the EWG on "sampling plans and numeric performance criteria for methods of analysis for total aflatoxins and ochratoxin A in spices." This work advanced to Step 8. It ensures that statistical sampling is used to obtain accurate results. India also contributed to the Code of Practice for the Reduction of Acrylamide in Foods, ensuring mitigation measures are practical for processors.
Codex Committee on Pesticide Residues (CCPR56) – Santiago
At the 56th session of the Codex Committee on Pesticide Residues (CCPR56) in Santiago, India chaired the development of "Guidelines for Monitoring the Stability and Purity of Reference Materials and Related Stock Solutions of Pesticides during Prolonged Storage." Advanced to Step 8, this document reduces the recurring costs associated with the procurement of certified reference materials, minimizes waste and ensures confidence in the reliability of pesticide residue analysis. India also provided monitoring data to set the Codex maximum residue limit (CXL) for tebuconazole in cumin at 0.9 mg/kg, ensuring the limit reflects real-world farming practices.
FAO/WHO Coordinating Committee for Asia (CCASIA23)
At the 23rd session of the FAO/WHO Coordinating Committee for Asia (CCASIA23), the Committee established an EWG chaired by India to review the Regional Code of Hygienic Practice for Street-Vended Foods in Asia. The revision aligns traditional practices with the "General Principles of Food Hygiene (CX 1-1969)" and "Guidelines for Food Hygiene Control Measures in Traditional Markets for Food (CXG 103-2024)," specifically addressing time and temperature controls in open-air markets.
Codex Committee on Spices and Culinary Herbs (CCSCH8) – Guwahati
At the 8th session of the Codex Committee on Spices and Culinary Herbs (CCSCH8) in Guwahati, India supported progress on standards for dried coriander seeds, vanilla, and large cardamom. On technical grounds, India opposed merging cassia and cinnamon into a single standard. This distinction is based on different physico-chemical profiles of the two spices, particularly regarding coumarin content, which requires separate safety evaluations.
Codex Executive Committee (CCEXEC89) & Codex Alimentarius Commission (CAC48) – Rome
Representing Asia at the 89th session of the Codex Executive Committee (CCEXEC89), India called for sustainable funding for scientific advisory bodies. The delegation also suggested using AI-based tools to speed up risk assessments to prevent delays in policy making after the Codex Trust Fund phases out.
The year ended at the 48th session of the Codex Alimentarius Commission (CAC48) in Rome, where India was re-elected to the Executive Committee 'as a member elected' on a geographical basis (Asia). The Commission adopted the standards for Fresh Dates and the Guidelines on Pesticide Reference Materials. From proposing new work on cashew kernels to providing data for global safety limits, India continues to be a consistent, science-based partner in global food safety.
