Abstract
This paper provides a technical overview of the regulatory advancements initiated by the Food Safety and Standards Authority of India (FSSAI) during 2025. Driven by socioeconomic shifts and the rapid expansion of the food ecosystem, these amendments represent a proactive alignment of national standards with contemporary scientific data and international benchmarks. By institutionalizing rigorous specifications across diverse categories, ranging from Refractive Index (RI) ranges in edible oils and microbiological safety in meat matrices to specific mineral density thresholds in packaged drinking water, FSSAI has strengthened the regulatory framework to mitigate emerging risks in the food supply chain. This paper details the scientific underpinnings of these amendments and their implications for enforcement, industry compliance, and public health nutrition.
Keywords: FSSAI; Food Regulation; Public Health Nutrition; Food Safety; Regulatory Amendments
1.0 Introduction
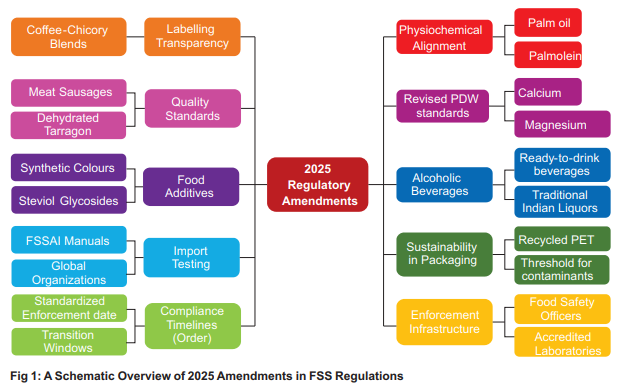
The Indian food consumption landscape is evolving faster than ever. The consumer's preferences for a more diversified food choice from traditional, innovative foods to packaged and convenience food is driven by socio economic and cultural transformation, urbanization and lifestyle. Recognizing the significant expansion of the food ecosystem and the heightened expectations of consumers, FSSAI has implemented a series of crucial regulatory updates in 2025, as shown in Fig 1. These were developed through a rigorous process of science-based scrutiny and multi-stakeholder consultation.
2.0 Transparency in Labelling Requirement to Empower Consumers
The heterogeneous nature of the beverage market necessitated more transparency in coffee-chicory blends to facilitate informed dietary choices. Since coffee and chicory possess distinct phytochemical profiles and vary significantly in caffeine concentration, the Food Safety and Standards (Labelling and Display) First Amendment Regulations, 2025, mandated precise Front-of-Pack (FOP) proportions. For instance, the regulations specify that instant coffee-chicory mixtures must contain a minimum coffee content of 51 percent by mass on a dry basis. This amendment addresses the requirement for consumer awareness regarding the percent mixture of coffee and chicory for better decision while purchasing.
3.0 Global Harmonization of Physicochemical Parameter of Edible Oils
The Refractive Index (RI) serves as a fundamental analytical parameter for verifying purity and preventing the use of cheaper substitutes. FSSAI aligned India's standards with Codex Alimentarius, the global benchmark for food safety, by amending the refractive index values for Palm Oil, Palmolein, Palm Kernel Oil, and Palm Superolein [Amendment 84, July 2025]. For Palm Oil, the RI range was revised from '1.4491-1.4552' to '1.454-1.456', while Palmolein was revised from '1.4550-1.4610' to '1.458-1.460'. Similar technical refinements were applied to Palm Kernel Oil and Palm Superolein. This alignment with Codex standards provides a robust verification mechanism for enforcement agencies to detect potential adulteration and maintain the chemical integrity of imported and domestic edible oils, thereby safeguarding the health of consumers and ensuring the authenticity of edible oils.
4.0 Quality Standards for Processed Foods
The establishment of specifications for meat sausages and dehydrated tarragon [Amendment 84, July 2025] addresses inherent vulnerabilities in processed food matrices. For meat sausages, the high surface-area-to-volume ratio of minced meat makes it highly susceptible to rapid microbial proliferation and lipid oxidation. This new regulatory framework establishes requirements for composition, thermal processing, and cold-chain storage to mitigate the risks of enteric pathogens such as Salmonella and Listeria monocytogenes. Similarly, standards for dehydrated tarragon aim to fill a void in current spice and herb regulations while improving product quality criteria and respond to its increasing utilization in the gourmet sector. Its standardization has benefited spice processors, food service enterprises, and premium exports. Since dehydrated botanicals are prone to moisture-induced microbial spoilage and the loss of bioactive volatile oils, FSSAI established standards for moisture content (not more than 10 percent) and volatile oil thresholds to ensure the retention of the phytochemical profile.
5.0 Revision in Standard of Packaged Drinking Water
The proposed amendment to the standards governing the quality of packaged drinking water [Amendment 84, July 2025], specifically targeting the Total Dissolved Solids (TDS) levels and permissible limits of vital minerals, such as calcium and magnesium, aim to not only ensure the safety and purity of bottled water but also enhance its nutritional value. By setting specific thresholds for calcium and magnesium content, FSSAI seeks to ensure that packaged drinking water serves as a supplemental source of essential minerals that contribute to human health. Calcium levels are now standardized between 10 to 75 mg/L and Magnesium between 5 to 30 mg/L.
6.0 Standardization of Food Additives, Colours, and Sweeteners
Revised provisions for "Food Colour - Preparation and Mixtures" and the expanded application of steviol glycosides address the safety and technological requirements of food additives [Amendment 84, July 2025]. Since synthetic colours are subject to strict Acceptable Daily Intake (ADI) thresholds, standardizing mixture preparation prevents accidental over-exposure. The natural, non-caloric sweetener extracted from stevia plants is gaining popularity. Historically, its application in certain beverage subcategories was restricted to ready-to-drink products and pre-mixes for ready-to-drink products only. This amendment allows steviol glycosides in a wider array of products within the beverage category while maintaining safety standards. This provides Food Business Operators with enhanced flexibility, opportunities for sugar-reduced innovations, and simplified reformulation of products.
7.0 Regulation for Innovative and Traditional Alcoholic Beverages
The Food Safety and Standards (Alcoholic Beverages) First Amendment Regulations, 2025, standardized a new category of Ready-to-Drink (RTD) alcoholic beverages, Mead, and Nitro Craft Beer. The new category of RTDs are beverages containing 0.5% to 15% alcohol by volume (ABV), created from spirits or any other alcoholic beverage as a base, mixed with permitted additives, flavors, fruit or vegetable juices, spices, herbs, with or without added sugar or caloric sweeteners, or salt, and may include carbonation. For carbonated alcoholic ready-to-drink beverages, the regulations mandate the use of Carbon dioxide to create effervescence, requiring a minimum of one volume of Carbon dioxide. The modifications facilitate increased innovation among RTD companies, microbreweries, and craft beverage manufacturers.
This amendment refines the classification of local liquors and provides recognition for traditional Indian liquors. By incorporating an official list of Indian liquors, the amendment acknowledges a wide range of indigenous alcoholic beverages, thereby bringing these products into the mainstream of the Indian market. This recognition aligns with the national vision of "Vocal for Local," while also contributing to enhanced livelihood opportunities for Food Business Operators (FBOs) engaged in the production of such traditional beverages. The standards have been framed with the objective of supporting and standardizing traditional alcoholic beverages commonly referred to as country liquors or Indian liquors based on their geographic origin, thereby preserving their unique identity, ensuring quality, and facilitating regulated market access.
In addition, the Second Amendment Regulations, 2025 revised the maximum permissible level of Esters expressed as ethyl acetate in fruit wines from "0.2" to "3.0" to align with the levels of naturally occurring esters.
8.0 Alignment of Testing Methodology and Report for Imported Food Articles
The Food Safety and Standards (Import) Regulations, 2017 mandated that notified and referral laboratories adhere to the testing methodologies prescribed by the Food Authority for the analysis of food samples, encompassing the requisite procedures and equipment. In instances where the Authority did not specify a particular method, laboratories were instructed to implement testing standards established by internationally recognized organizations such as the Codex Alimentarius Commission, the International Organization for Standardization (ISO), or guidelines provided by the Directorate General of Health Services (DGHS). To comply with the Food Safety and Standards Rules, 2011, and to clarify the procedures for testing imported articles of food, the Food Safety and Standards (Import) First Amendment Regulations, 2025 have been notified.
This amendment gives flexibility by necessitating the utilization of the FSSAI's official Manuals of Methods of Analysis, which is periodically updated. If a required method is absent from these manuals, laboratories may adopt a validated method endorsed by esteemed global organizations, including the Association of Official Agricultural Chemists (AOAC), ISO, Pearson's, Jacob, International Union of Pure and Applied Chemistry (IUPAC), Food Chemicals Codex, BIS, Codex Alimentarius, Woodmen, Winton–Winton, Joslyn, or any other internationally recognized regulatory agencies. This amendment harmonizes the import testing framework with the Food Safety and Standards Rules, 2011, assuring consistency in testing protocols for imported samples, while allowing laboratories to implement contemporary, scientifically established methodologies as needed.
9.0 Sustainability in Packaging: rPET Guidelines
Taking into account the environmental impact of food packaging and sustainability, FSSAI permitted the use of recycled Polyethylene Terephthalate (rPET) for food contact materials [Amendment 3, March 2025]. To address the risk of chemical migration from recycled plastic, the Authority notified the Guidelines for acceptance of recycled Polyethylene terephthalate (PET) as Food Contact Material (FCM-rPET) to serve as a reference for FBOs and ensure compliance with the national standards or regulations as applicable to such materials. These guidelines ensure that contaminants are reduced below the threshold levels, facilitating the circular economy without compromising the chemical safety of the packaged food or the health of the consumer.
10.0 Policy Adopted for Ease of Doing Business
The challenges faced by FBOs are primarily related to the implementation of the regulations, required operational changes, and associated costs. Additionally, the use of pre-printed packaging materials pose obstacles in ensuring compliance with these amendments. Recognizing these issues, FSSAI took a significant step to address the challenges faced by the FBOs and provide them with a predictable and efficient framework for compliance with food labelling regulations. A decision that would promote ease of doing business, FSSAI has fixed the date of enforcement of labelling amendments specified under the Food Safety and Standards (Labelling and Display) Regulations, 2020, starting from July 1st every year subject to minimum of 180 days from the date of notification for the amendments related to FSS (Labelling and Display) Regulations and also for any change in labelling specified in other FSS Regulations [Order, January 2025].
11.0 Analytical and Enforcement Infrastructure Strengthening
The efficacy of these regulations is linked to the strength of the nation's enforcement and laboratory network. Throughout 2025, FSSAI significantly expanded its operational framework through a continuous series of Gazette Notifications. This included the strategic appointment of Central Food Safety Officers (Jan, Feb, June, July, Aug 2025) and specialized Food Safety Officers for Railways (Aug, Sept 2025), Airports, and Seaports (July, Nov 2025). These appointments were complemented by two specific enforcement drives (Aug, Nov 2025) involving 266 additional Food Safety Officers authorized to monitor the safety and distribution of fortified rice kernels.
To bolster technical capacity, FSSAI notified the appointment of Food Analyst personnel under Section 45 (Jan, Sept 2025) and expanded the Equivalent Qualification criteria for Food Safety Officers in March 2025. Governance was further strengthened through the notification of the Central Advisory Committee (CAC) in October 2025.
To ensure analytical precision, the Authority expanded the laboratory network to 260 National Accreditation Board for Testing and Calibration Laboratories (NABL) accredited laboratories and 24 Referral Food Laboratories through updated notifications in June and November 2025. Furthermore, 305 Mobile Food Testing Laboratories (Food Safety on Wheels) have been deployed under the Strengthening of Food Testing Laboratories (SoFTeL) scheme to provide rapid screening and consumer awareness at the field level.
References
Amendment 3. (2025, March 28). Food Safety and Standards (Packaging) First Amendment Regulations, 2025. Food Safety and Standards Authority of India.
Amendment 84. (2025, July 10). Food Safety and Standards (Food Products Standards and Food Additives) First Amendment Regulations, 2025. Food Safety and Standards Authority of India.
Food Safety and Standards Authority of India. (2025, January 3). Order No. REG-17/2/2022-Regulation-FSSAI: Decision taken in the 45th Food Authority Meeting regarding timeline for the compliance of amendments in labelling provisions.
