1.0 Introduction
Under Section 18 of the FSS Act, the FSSAI is responsible for protecting consumer health and ensuring fair trade practices. This includes preventing deceptive or fraudulent activities that could mislead or harm the public. Section 16 further empowers the FSSAI to set specific regulations for food labeling, covering areas like health claims, nutritional information, and special dietary uses.
To support these goals, the Food Safety and Standards (Advertising and Claims) Regulations, 2018 were established. These rules ensure that food advertisements are honest and hold businesses accountable for the claims they make, ultimately safeguarding consumer interests.
2.0 What are Nutrition and Health Claims?
A claim is any "message" or "representation" that manufacturers use to suggest that a food has certain qualities. This can include text (like "gluten-free," "apple flavor," or "low fat") and pictures, graphics, and symbols. For example, a product could claim it was 'vegan' on the label whilst another may use an image of the gut which could imply that the product has a positive effect on gut health. Not all claims have strict requirements that need to be followed, and in these cases, whether the claim is permitted comes down to if it is considered misleading under relevant legislation. However, there are specific types of claims (as shown in Fig 1), including nutrition and health claims that have to meet set criteria before being used for a product. But firstly, what exactly do we mean by nutrition and health claims?
- Nutrition claims: The clue is in the name — these relate to a specific nutrient (e.g. fat, sugar, protein, fibre) and suggest a beneficial effect (e.g. low fat, sugar-free, source of protein, high fibre).
- Health claims: These go one step further — they make a statement that connects a nutrient/food with a positive effect on health (e.g. vitamin D contributes to the normal function of the immune system; plant sterols contribute to the maintenance of normal blood cholesterol levels).
Types of Claims (Fig 1)
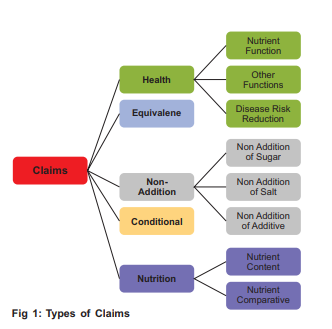
3.0 How Prevalent are Nutrition and Health Claims?
In 2016, a five-country study in Europe (including the UK) found that out of 2,034 food and drinks sampled, 26% had at least one claim; of which, 64% were nutrition claims, and 29% were health claims (Coates et al., 2024). Of the claims, 40% related to processes involved in breaking down food, converting it to energy, and regulating the body's internal environment. Among the five countries studied, the UK had the highest proportion of products with at least one claim (35%).
A study published earlier this year reviewed the prevalence and compliance of products bearing claims online in Great Britain for dairy and alternatives, fruit juices and teas. In total, 28% of products displayed health claims, although not all claims (6–10%) displayed were compliant with the relevant legislation that is in place to regulate their use (FSSAI, 2018a).
4.0 Concept of A "Claim" in Regulatory Context
A claim is defined broadly as any representation — written, oral, audio, or visual — that states, suggests, or implies that a food has particular qualities relating to origin, nutritional properties, nature, processing, composition, or other characteristics (FSSAI, 2018b). This expansive definition ensures that not only explicit statements but also implied messages, visuals, and comparative references fall within regulatory oversight.

Claims are regulated because they may create health halos, exaggerate benefits, or obscure nutritional risks if not properly controlled. Consequently, the regulations classify claims into distinct types, each with specific conditions and evidentiary requirements.

4.1 Nutrition Claims
Nutrition claims are the most frequent type of claim and are relatively straightforward. According to Regulations 2(1)(l) and 5, these claims focus solely on a food's nutritional properties, such as its calorie count or specific nutrient levels.
4.1.1 Nutrient Content Claims
A nutrient content claim describes the level of a nutrient present in a food, either directly or indirectly. Examples include "low fat," "high fibre," or "source of protein" (FSSAI, 2018c). It is absolute in nature; no comparison with another food is required and should strictly follow quantitative thresholds specified in Schedule I.
Example:
- "High in dietary fibre" may only be used if the product contains at least 6 g fibre per 100 g (solids).
4.1.2 Nutrient Comparative Claims
A nutrient comparative claim compares the nutrient level or energy value of one food with another similar food (FSSAI, 2018d).
- Comparison must be between similar foods or different versions of the same food.
- Minimum percentage differences are prescribed (e.g., 30% for macronutrients, 10% RDA for micronutrients).
- The reference food and magnitude of difference must be clearly disclosed.
Example:
- "30% less sugar than regular biscuits."
4.1.3 Equivalence Claims
Equivalence claims suggest parity between the nutrient content of two foods (FSSAI, 2018e).
Example:
- "Contains as much calcium as a glass of milk."
Such claims are permitted only if both foods qualify as a source of the nutrient and equivalence is demonstrated on a per-100 g or per-100 ml basis.
4.2 Non-Addition Claims
Non-addition claims communicate the absence of an ingredient or additive that consumers might normally expect in a food (FSSAI, 2018f).
4.2.1 Non-Addition of Sugars
Claims such as "no added sugar" are permitted only when no sugars or sugar-containing ingredients have been added, and sugars have not been generated through processing methods. Where naturally occurring sugars are present, the label must state "CONTAINS NATURALLY OCCURRING SUGARS."
Example:
- Unsweetened fruit juice labelled as "No added sugar – contains naturally occurring sugars."
4.2.2 Non-Addition of Sodium Salts
Claims like "no added salt" are permitted only if no sodium salts or salt-substituting ingredients are used.
4.2.3 Non-Addition of Additives
Claims like "no preservatives added" are only permitted if the additive was not directly included or present in any of the ingredients. Furthermore, businesses cannot use this claim if they have simply replaced a standard preservative with another ingredient that performs the same function.
4.3 Health Claims
Health claims are more complex and stringently regulated because they link food consumption with health outcomes (FSSAI, 2018g).
Health claims are subdivided into three principal categories:
4.3.1 Nutrient Function Claims
These claims describe the physiological role of a nutrient in normal growth, development, or body functions.
Example:
- "Calcium is needed for maintenance of normal bones."
Such claims must be supported by generally accepted scientific evidence and the food must qualify as a source or rich source of the nutrient.
4.3.2 Other Function Claims
These claims provide specific benefits of foods or their components that go beyond basic nutrition by supporting natural biological or physiological functions.
Example:
- "Beta-glucans help reduce the rise in blood glucose after a meal."
While these claims do not directly mention reducing the risk of a specific disease, they must be backed by strong scientific evidence to be permitted.
4.4 Reduction of Disease Risk Claims
These claims suggest that consumption of a food reduces a risk factor for a disease, not the disease itself (FSSAI, 2018h).
Example:
- "Diets low in sodium may help reduce the risk of high blood pressure."
Conditions:
- Must not be interpreted as prevention or cure claims.
- Must specify context of total diet and, where required, number of servings.
- Only claims listed in Schedule III are permitted without prior approval.
4.5 Claims Related to Healthy Diets and Dietary Guidelines
Any claims describing food as part of a "healthy," "balanced," or "wholesome" diet must follow the specific recommendations outlined in the Indian Council of Medical Research (ICMR) Dietary Guidelines (FSSAI, 2018i).
Importantly, a single food product can never be advertised as inherently "healthy" all by itself.
Example:
- "Part of a balanced diet when consumed alongside fruits, vegetables, and regular physical activity."
Ingredient-Led vs. Product-Led Claims
Health claims could be Ingredient Led or Product Led:
- Ingredient-led claim example: "Probiotics supports good gut health." — Ingredient-led claims may be made based on current relevant scientific substantiation and to provide sufficient evidence on the type of claimed effect and the relationship to health as recognized by generally accepted scientific review of the data.
- Product-led claim example: "Product A supports good gut health." — Product-led claims shall be based on statistically significant results from well-designed human intervention studies, conducted by or under guidance of established research institutions, in line with the principles of GCP (Good Clinical Practices) and peer reviewed or published in a peer reviewed reputed scientific journal.
4.6 Conditional and Descriptive Claims
Conditional claims include the use of descriptors such as "natural," "fresh," "pure," "traditional," or "original" (FSSAI, 2018j). These terms are strictly defined and permitted only when objective criteria are met.
Example:
- "Made from natural ingredients" (permitted for composite foods, whereas "natural food" is not).
4.7 Prohibited Claims (FSSAI, 2018k)
The following claims are prohibited under the regulations:
- No claims shall be made which refer to the suitability of the food for use in the prevention, alleviation, treatment or cure of a disease, disorder or particular physiological condition unless specifically permitted under any other regulations made under Food Safety and Standards Act, 2006 (34 of 2006).
- There shall not appear in the label of any package, containing food for sale the words "recommended by the medical or nutrition or health professionals" or any words which imply or suggest that the food is recommended, prescribed, or approved by medical practitioners or approved for medical purpose.
- No product shall claim the term "added nutrients", if such nutrients have been added merely to compensate the nutrients lost or removed during processing of the food.
- Foods for special dietary uses or foods for special medical purposes shall not carry a claim unless specifically permitted under any other regulations made under Food Safety and Standards Act, 2006 (34 of 2006).
- Claims which do cause doubt or suspicion about the safety of similar food or which may arouse fear shall not be made.
- No health claims shall be made for foods that contain nutrients or constituents in quantity that increase the risk of disease or an adverse health-related condition.
- No advertisements or claims for articles of foods shall be made by any food business operator that undermines the products of any other manufacturer for the purpose of promoting their products or influencing consumer behaviour.
Comparative Perspective and Regulatory Rationale
The regulatory architecture reflects a graduated risk approach:
- Nutrition claims are low-risk and standardised.
- Non-addition and conditional claims carry moderate risk of consumer misinterpretation.
- Health and disease risk reduction claims pose the highest risk and therefore attract the strictest scrutiny.
| DO's | DON'T's |
|---|---|
| Truthful, Unambiguous, Meaningful, Help consumers comprehend information | Misleading |
| Specify the number of servings of the food per day for the claimed benefit | Do not encourage or condone excess consumption of a particular food |
| Claim should be scientifically substantiated by validated methods | Do not suggest against balanced and varied diets |
| Characterize or Quantify ingredients that is the basis for the claim | No claim on promotion, sale, supply, use and consumption of articles to be made using FSSAI logo |
| All disclaimers to be legible | Do not undermine healthy lifestyle |
| Advertisements not to portray/promote foods as Meal replacement unless specifically permitted |
5.0 Conclusion
The Food Safety and Standards (Advertising and Claims) Regulations, 2018 establish a framework for classifying food claims, with each category governed by specific definitions, nutrient limits, and scientific substantiation requirements. Understanding the distinctions between nutrition, health, non-addition, conditional, and prohibited claims is essential for regulators and food businesses. More information is available on the Food Safety and Standards website.
References
Coates, E., Pentieva, K., & Verhagen, H. (2024). The prevalence and compliance of health claims used in the labelling and information for pre-packed foods within Great Britain. Foods, 13, 539. https://doi.org/10.3390/foods13040539
Food Safety and Standards Authority of India. (2018a). Food safety and standards (advertising and claims) regulations, 2018: Regulation 2(1)(d). FSSAI.
Food Safety and Standards Authority of India. (2018b). Food safety and standards (advertising and claims) regulations, 2018: Regulation 2(1)(f) and Regulation 5(6). FSSAI.
Food Safety and Standards Authority of India. (2018c). Food safety and standards (advertising and claims) regulations, 2018: Regulation 2(1)(h) and Regulation 7. FSSAI.
Food Safety and Standards Authority of India. (2018d). Food safety and standards (advertising and claims) regulations, 2018: Regulation 2(1)(h)(iii), Regulation 7(3), and Schedule III. FSSAI.
Food Safety and Standards Authority of India. (2018e). Food safety and standards (advertising and claims) regulations, 2018: Regulation 2(1)(l)(i), Regulation 5(3), and Schedule I. FSSAI.
Food Safety and Standards Authority of India. (2018f). Food safety and standards (advertising and claims) regulations, 2018: Regulation 2(1)(l)(ii) and Regulation 5(4). FSSAI.
Food Safety and Standards Authority of India. (2018g). Food safety and standards (advertising and claims) regulations, 2018: Regulation 2(1)(m) and Regulation 6. FSSAI.
Food Safety and Standards Authority of India. (2018h). Food safety and standards (advertising and claims) regulations, 2018: Regulation 8. FSSAI.
Food Safety and Standards Authority of India. (2018i). Food safety and standards (advertising and claims) regulations, 2018: Regulation 9 and Schedule V. FSSAI.
Food Safety and Standards Authority of India. (2018j). Food safety and standards (advertising and claims) regulations, 2018: Regulation 10. FSSAI.
Food Safety and Standards Authority of India. (2018k). Food safety and standards (advertising and claims) regulations, 2018: Regulation 10. FSSAI.
