Abstract
Food contact substances (FCSs) refer to those materials which come into contact with food at some or the other stage of food handling (including processing, packaging, storage, or preparation). This includes materials used throughout the food supply chain, such as processing equipment, adhesives, household utensils, printing inks, coatings, stabilizers, colorants, etc. A major issue that arises when food contact materials (FCMs) come in contact with food material is the migration of the particles of FCMs into food. The modern-day packaging materials such as synthetic polymers, bio-based plastics, multilayer laminates along with recycled materials involve heavy usage of chemicals during its manufacturing or processing. Therefore, these materials contain huge amounts of intentionally added substances (IAS) and non-intentionally added substances (NIAS), which may migrate from these materials into the food product that comes in contact with these materials. The recent studies conducted on over 3,600 chemicals used as FCSs have been found to have negative impacts on the human body. The chemicals have been found to be the endocrine disruptors, metabolic disruptors, and carcinogenic as well. The enhancement in the structural complexity of packaging, leading to the shift of industries towards combined sustainability and recycling, has further added newer challenges, including scientific, analytical, and regulatory. This review summarizes the latest updates on different varieties of FCM, migration pathways, health hazards, latest regulations to administer its usage, and following research thrusts. Based on these studies, the article also highlights the urgent need for a unified global standard, better assessment of NIAS and development of packaging materials which are comparatively safer and innovative.
Keywords: Carcinogenic; Chemical Migration; Food Contact Materials; Packaging Safety; Photoinitiators
1.0 Introduction
Food packaging is required to store food fresh throughout long durations of time, safeguard food against physical, chemical, and microbial contamination. This has the effect of increasing food shelf life. The food wrapping that comes in contact with the food is a barrier that prevents entry of moisture, air, light, and other degrading elements into a food and contributes to rotting of food. This dichotomy of protection in case of possible contamination is an inherent element that is needed in the ongoing debates concerning the safety of food contact materials (FCMs). The FCMs are those that come in direct or indirect contact with food (Groh et al., 2020). They include plastics, paper & board, metals, glass, ceramics, elastomers, adhesives, inks, and new options including bio-based polymers and recycled plastics. These materials have a wide range of chemical compounds, such as monomers, oligomers, plasticizers, stabilizers, colorants, catalysts, and processing aids. The way these chemicals get into food depends on the chemistry of the packaging, the environment, the qualities of the food, how long it is stored, and the temperature.
These packaging materials harbor various constituent chemicals — monomers, stabilizers, plasticizers, colorants, and processing aids, which can seep out into the food. The rate at which this migration occurs hinges critically on factors such as the packaging's chemical composition, ambient storage temperature, the food's specific properties, and the overall duration of contact. Driven by how we consume products today, packaging technology has evolved rapidly. We now see everything from multilayered laminates and nanocomposites to "smart" active packaging and compostable films. While these breakthroughs make packaging much more functional, they also introduce a new wave of potential "migrants" — chemicals that leak into our food. This includes non-intentionally added substances (NIAS), which are essentially accidental byproducts created during manufacturing, recycling, or chemical degradation (Thoden et al., 2023).
The rise of modern consumer habits has led to the creation of innovative packaging technologies such multilayer flexible laminates, nanocomposites, high-barrier coatings, active and intelligent packaging, biodegradable films and recycled polymer systems. These new ideas make things work better, but they also bring in new potential migrants, such as unknown NIAS that arise through degradation, manufacturing processes, impurities, and recycling pollutants. Recent (2024) biomonitoring investigations detected more than 3,600 food-contact-related compounds in human blood, urine, and serum (Fig 1). Phthalates, bisphenols, mineral oil hydrocarbons, photoinitiators, amines, and oligomers are all linked to endocrine problems, reproductive damage, cancer, metabolic problems, and neurotoxicity. This scary evidence has made calls for better testing methodologies, revised rules, and clearer risk assessment frameworks even stronger.
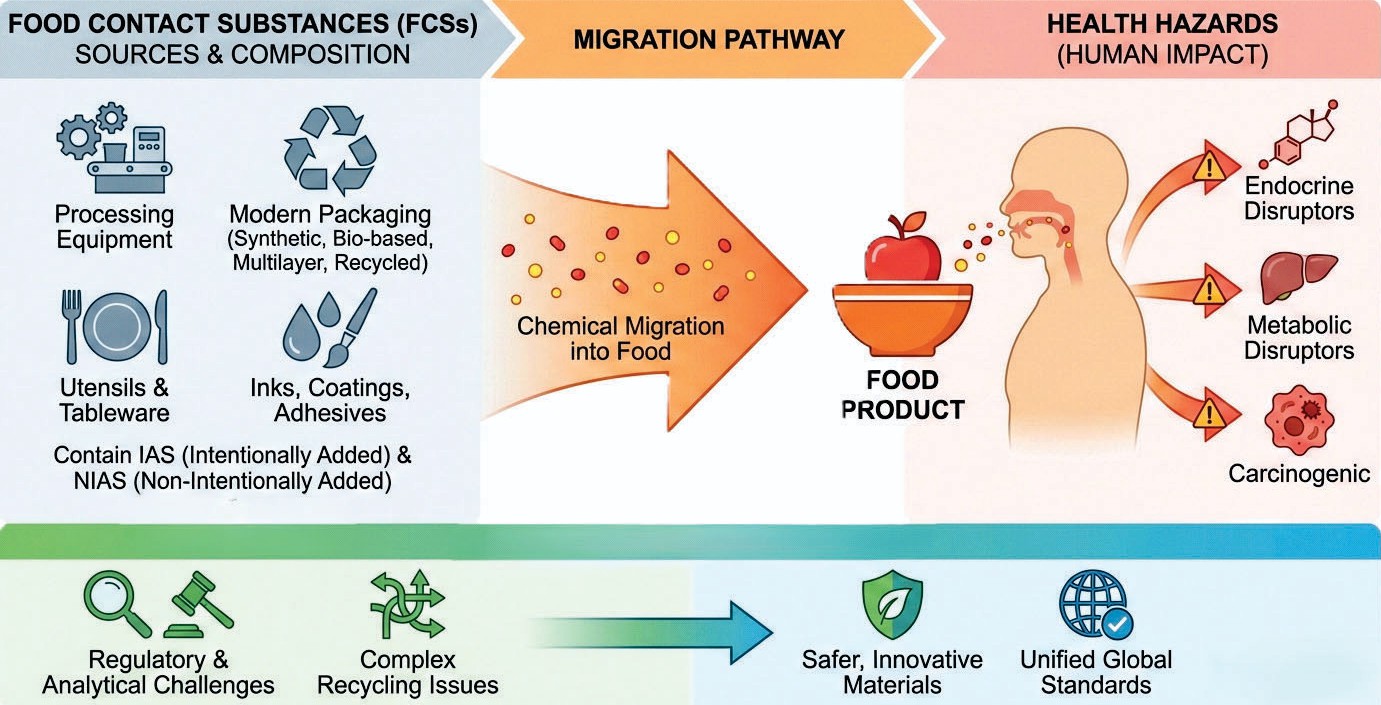
Fig 1: Food Contact Chemicals Detected in Human Samples
2.0 Types of Materials Used in Food Contact Applications
There are thousands of materials used for packaging and processing food worldwide. The safety of these materials depends on the chemicals they are made of and how easily their parts can move about.
| Category | Examples |
|---|---|
| Plastics / Polymers | PE, PP, PET, PVC, PS, PC, Nylon, EVOH, ABS, etc |
| Paper & Paperboard | Kraft paper, parchment paper, paperboard, molded fiber |
| Coated & Laminated Paper | PE-coated paper, aluminum-coated, biopolymer-coated |
| Metals | Aluminum, tinplate, stainless steel |
| Glass | Soda-lime glass, borosilicate glass |
| Ceramics & Porcelain | Glazed ceramics, stoneware |
| Elastomers / Rubbers | Silicone, nitrile rubber, EPDM, natural rubber |
| Biodegradable Biopolymers / Compostable | PLA, PHA, starch-based polymers, cellulose films |
| Composites / Multilayer Materials | Plastic-plastic laminates, plastic-aluminum, paper–plastic |
| Textiles | Cotton, synthetic fibers |
| Inks, Coatings & Adhesives | UV inks, solvent-based inks, lacquers, epoxy coatings, laminating adhesives |
| Wood & Bamboo | Untreated or treated wood, bamboo composites |
3.0 Migration Mechanisms
Migration refers to the movement of chemicals from FCMs into food. It occurs by a combination of both physical and chemical processes; these may include diffusion, partitioning, evaporation, and penetration. The primary mechanism by which things occur is diffusion and is exerted wholeheartedly by items such as the molecular structure of the polymer, the size and polarity of additions, the level of crystallinity and external factors such as temperature and the duration things are in contact with one another. Temperature is also a very significant variable; a slight increase in it can significantly accelerate the flow of molecules in the substance, accelerating the rate of migration.
Partition coefficients determine the distribution of a substance when it diffuses out of the material, and it is between the FCM and the food matrix. These coefficients tell us how the migrant divides into two phases at equilibrium. This partitioning is highly dependent on the composition of the meal. The lipophilic (fat-loving) substances are more readily absorbed into fatty foods, and consequently, plasticizers, antioxidants and other additives that are hydrophobic relocate more. Conversely, watery or acidic meals are likely to get hydrophilic substances, such as some monomers or ionic degradation products (Groh et al., 2020).
The latest scientific researches have contributed to the improved understanding of the migration processes and have identified multiple new avenues beyond the mechanisms of diffusion, which are controlled by the diffusion. Migration is of many varieties, such as set-off migration, which occurs when the chemicals migrate between printed or coated surfaces when materials have been stacked or rolled, gas-phase migration, when volatile compounds move through the headspace without contact, migration by contact condensation, when water droplets or steam dissolve to carry migrants to the food, and migration through multilayer structures, when substances move through barrier layers previously understood to be impermeable. All these findings underscore the complexity of migratory behavior and the need to have sophisticated analysis methodologies to the modern food packaging system (Begley, 2020).
4.0 Analytical Methods for Identifying and Quantifying Migrants
To locate FCM migrants, especially NIAS, you need powerful analytical tools. Overall migration considers the total amount of non-volatile chemicals that are transferred in the food simulants. GC-MS, HS-GC, HPLC-MS/MS, UHPLC-QTOF-MS, and ICP-MS are all tests for specific migration. Recent (2025) investigations have identified benzene derivatives, amines, nitrites, and oligomers in packaged goods, thus underlining the diversity of contaminants. The NIAs are derived from things such as impurities, byproducts, deterioration, and pollutants that are recycled. It is hard to find them because we do not know what it is, and there are no standards and there is no sufficient or enough toxicological data. To fill these gaps, high resolution mass spectrometry and non-targeted screening are being used in recent times (Nerín et al., 2023).
5.0 Regulatory Frameworks
The FCMs have a lot of regulations, worldwide, and they exist to ensure the safety of people. Nonetheless, these regulations differ greatly across regions of the world, as follows:
5.1 European Union (EU)
The European Union operates one of the most comprehensive and science-based frameworks for food contact materials. The cornerstones of EU regulations are:
- Framework Regulation (EC) No 1935/2004 – Establishes general safety principles for all materials and articles intended to come into contact with food.
- Requires that materials do not transfer constituents to food in quantities that could endanger human health, change food composition, or alter sensory qualities.
- Mandates traceability and labeling throughout the supply chain.
This regulation is supported by more specific measures, including:
- Commission Regulation (EU) No 10/2011 — Governs plastic materials and articles, providing a positive list of authorized substances, specific migration limits (SMLs), and overall migration limits (OMLs).
- Regulation (EC) No 2023/2006 — Establishes Good Manufacturing Practice (GMP) requirements for all FCMs.
- Specific Directives or Measures — Cover materials such as regenerated cellulose film (2007/42/EC), ceramics (84/500/EEC), active and intelligent packaging (450/2009), and recycled plastics (2022/1616).
5.2 United States
In the United States, the Food and Drug Administration (FDA) regulates FCMs under the Federal Food, Drug, and Cosmetic Act (FD&C Act). Key regulatory mechanisms include:
- Title 21 of the Code of Federal Regulations (CFR) – Lists authorized substances for specific material categories (e.g., 21 CFR 177 for polymers, 21 CFR 175 for adhesives, and 21 CFR 178 for adjuvants and production aids).
- Food Contact Notifications (FCN) — Allow manufacturers to obtain FDA clearance for new substances used in FCMs. Once an FCN is approved, it applies exclusively to the notifier and specific conditions of use.
- Threshold of Regulation (ToR) exemptions — Permit use of substances with negligible migration and risk.
- Good Manufacturing Practices (21 CFR 174.5) — Mandate sanitary and safe production conditions for all food contact articles.
The US approach is substance-based and relies heavily on toxicological evaluation, exposure assessment, and migration modelling. It provides flexibility for innovation, but places responsibility on manufacturers to ensure safety under intended use conditions.
5.3 Canada
Health Canada's Food Directorate regulates FCMs under the Food and Drugs Act. While Canada has no single comprehensive list equivalent to the EU's positive list, it maintains an internal database of reviewed and approved substances. Manufacturers may voluntarily submit pre-market notifications to obtain safety evaluations for new materials. Canada's approach aligns closely with the US FDA model and recognizes many FDA clearances as supportive evidence.
5.4 Asia-Pacific Region
5.4.1 China
The National Health Commission (NHC) administers a well-defined system under:
- GB 4806.1-2016: General safety standard for FCMs.
- GB 9685-2016: Positive list of additives for food contact materials.
- GB 4806.7-12: Series for specific materials such as plastic, paper, rubber, and coatings.
- Compliance involves meeting migration limits, labeling requirements, and local testing.
5.4.2 Japan
Governed by the Food Sanitation Act, amended in 2020 to introduce a positive list system for synthetic resins, aligning Japan more closely with EU and FDA approaches. Testing and documentation are required for all new materials.
5.4.3 India
Food Safety and Standards Authority of India (FSSAI) oversees packaging safety under the Food Safety and Standards (Packaging) Regulations, 2018, covering plastics, metals, glass, paper, and multilayer laminates.
5.5 Latin America
Regulatory systems in Brazil, Mexico, Argentina, and Chile are based largely on Mercosur Resolutions, particularly:
- Mercosur GMC Resolution No. 03/92 — General principles for FCMs.
- Mercosur GMC Resolution No. 32/07 — Technical regulation for plastic materials and articles.
These are harmonized with EU and FDA concepts, including positive lists and migration testing requirements. Brazil's ANVISA enforces compliance at the national level.
As indicated above, Plastics Regulation (EU) No. 10/2011 is more detailed on what is permitted and what is not permitted with a list of approved compounds, maximum level of migration, tests, and multilayer specifications. Recently, this has become more difficult due to the increasing concerns with the use of substances that are increasingly of concern, such as styrene, the bisphenol analogues (such as BPA, BPS and BPF), titanium dioxide nanoparticles, and printing inks chemicals. The reason behind this is that the emerging scientific studies are proving that such substances could be harmful.
The United States government has numerous means by which the USFDA monitors FCSs. The Food Contact Notification (FCN) program allows individuals to verify the safety of new compounds before the market. Rules on indirect additives are contained in Title 21 CFR, and there are exemptions of Threshold of Regulation (TOR) concerning items that are very unlikely to be exposed. This dynamic, risk-based framework has allowed the sector to have a great deal of accountability to demonstrate that novel concepts are secure.
The Bureau of Indian Standards (BIS) and the Food Safety and Standards Authority of India (FSSAI) are the two major bodies that ensure that regulations are adhered to in India. The FSSAI establishes a threshold of the total migration, particular migration of heavy metals, primary aromatic amines, phthalates, and monomers. The BIS, conversely, formulates standards for plastics and polymeric materials used in packaging, printing inks standards, such as prohibition of toluene-based inks and unfortunate colorants. The regulatory framework in India is being upgraded to global standards. Although considering realistic scenarios, demographic diversity across rural and urban areas in Indian context, there are challenges to implement enforcement.
The most significant issue that affects all states is the occurrence of NIAS compounds that enter the material accidentally as an impurity or as the byproduct of reaction or degradation or in recycled feedstock. The NIAS are not easy to address, because many cases cannot be fully identified or measured using the current specific analytical methods. Experts recommend that hazard identification and risk assessment should be conducted at the level of the entire material rather than those known individual substances. The reason is that toxicity may arise with known and unknown chemicals and the combination of mixtures may be synergistic or cumulative.
On the global arena, international fragmentation remains a huge issue to the safe international trade and uniform compliance and in view of different technical barriers to trade (TBT) notifications by countries. Manufacturers operating in more than one market are not sure of what to expect, due to the variance in substance authorization list, circumstances of migration testing, NIAS standards, and recycled plastics criteria. Although Codex Alimentarius and industry associations are being involved in it, still no global standards of FCMs can be found that are identical across the board. It is particularly the case with NIAS, recycling or chemical upcycling of advanced materials, and polymers. This demonstrates the significance of having safety frameworks that work across borders and regulatory science working across borders (Franz & Welle, 2022).
6.0 Potential Migrants and Health Concerns
Numerous scientific sources are proving that numerous chemicals contained in FCMs are not only known to be harmful but becoming known to be harmful, which casts doubt on the safety of the consumers. Some of the most studied migrants are plasticizers, especially phthalates. They are applied to make the polymers more flexible. Much of the toxicology study revealed that they have the capacity to interfere with hormonal signalling to disrupt the normal functioning of hormones and impair reproductive development and metabolic diseases. Several of the more risky phthalates have been stricter or even banned by regulatory agencies across the globe, but remain because of antique materials, incidences of cross-contamination, and the use of alternative plasticizers, the safety profile of which is not fully known.
Another large category of items that concerns people is bisphenols, which is utilized in polycarbonate plastics, epoxy can finishings and thermal papers. BPA, BPS, BPF, and other structural equivalents are also examples of bisphenols. They are associated with reproductive diseases, neurodevelopmental effects, immune system alteration and potential metabolic changes such as obesity and diabetes as these substances contain estrogenic and anti-androgenic qualities, assuming the role of hormones and inhibiting them. Although much criticism and restriction has been placed on BPA, its substitutes which are often marketed as BPA-free are now showing the same endocrine-disrupting properties. This exhibits a process of ill substitution.
Mineral oil hydrocarbons (MOH) are another complicated collection of pollutants that can come from inks, adhesives, recycled cardboard, and lubricants used in processing. In this group, mineral oil aromatic hydrocarbons (MOAH) are especially worrisome since some parts of them may induce mutations or cancer, and there is proof that they build up in human tissues such the liver, spleen, and lymph nodes. Even if regulators are paying more attention to MOH, it is still hard to analyze them because their compositions are so complicated. Photoinitiators, which are used in UV-cured printing inks and coatings, can also be harmful to health. Several frequently utilized photoinitiators have been recognized as genotoxic, cytotoxic, or endocrine-active, and exhibit a tendency to move into food, particularly under insufficient curing conditions or when utilizing low-barrier materials such as paper and thin films. High-profile cases involving 4-methylbenzophenone and ITX have shown that there are holes in the control mechanisms and that printing processes need to be watched more closely.
The most difficult group is NIAS, which includes a huge number of impurities, degradation products, chemical intermediates, oligomers, and contaminants that come from raw materials, additives, or recycling processes. Researchers have found thousands of unknown compounds in blood, urine, and tissues thanks to improvements in analytical chemistry and human biomonitoring. Many of these chemicals may come from food packaging and other things we use daily. Because we do not know much about NIAS, we do not know much about their toxicological profiles, interactions, or cumulative effects. This makes typical risk assessment methods very difficult. These results show how difficult it is to figure out how safe chemicals are in FCMs, and they stress the need for more toxicological tests, better analytical methods, and stricter rules.
7.0 Conclusion
Safety concerns have been compounded by rapid changes in packaging technology and the need to be environmentally friendly. Recycled and bio-based materials will be more environmentally friendly, but they usually generate more NIAS due to plant pollutants, byproducts of fermentation, and old toxins. Few of the 12,000 or so known food-contact compounds have been rigorously tested for toxicity. Many testing labs lack the high-resolution mass spectrometry that is required to make a robust NIAS evaluation. The primary issues include the fact that NIAS standard is not rigid enough, International SMLs are not universal, and a control over e-commerce packaging is also ineffective. The FAO (2024) recommends an all-inclusive approach that considers human health, environmental effects, microplastic pollution, and the environmental impacts of packaging chemicals.
Chemical migration of FCMs continues to be a significant food safety issue in all parts of the world. The regulatory frameworks are essential to safety and the introduction of new materials, sustainability, and improved container designs complicate everything significantly. The discovery of thousands of chemicals related to packaging in human biomonitor specimens demonstrates the significance of the presence of the updated regulatory procedures, improved analysis techniques, and safer materials design.
Future goals are the development of non-targeted analytical methods, NIAS rules are to be more powerful, global harmonization is encouraged, computational toxicology is used and the cooperation between industry, researchers and politicians is promoted in the world. The health of consumers will be at risk and new materials science, analytical chemistry, and food safety regulations will be necessary together with the coordinated efforts on the global level.
| Parameter | Limit |
|---|---|
| Overall Migration (OML) | 10 mg/dm² or 60 mg/kg |
| BPA (SML) | 0.05 mg/kg |
| Phthalates (SML range) | 0.3–1.5 mg/kg |
| PAAs | 0.01 mg/kg (or ND for carcinogens) |
| Heavy Metals (Pb) | < 0.01 mg/kg |
| VCM | 0.01 mg/kg |
| TOR Exposure Limit | 0.5 μg/person/day |
References
Begley, T. H. (2020). Migration of components into food: Principles and regulatory approaches. In K. Barnes, R. Sinclair, & D. Watson (Eds.), Chemical migration and food contact materials (2nd ed.). Woodhead Publishing.
Bureau of Indian Standards. (2016–2024). Standards for plastics, paper, metal containers, and printing inks for food-contact applications. BIS.
European Commission. (2006). Commission Regulation (EC) No 2023/2006 on good manufacturing practice for materials and articles intended to come into contact with food. Official Journal of the European Union.
European Commission. (2011). Commission Regulation (EU) No 10/2011 on plastic materials and articles intended to come into contact with food. Official Journal of the European Union.
European Food Safety Authority Panel on Food Contact Materials, Enzymes and Processing Aids (CEP). (2023–2024). Scientific opinions on bisphenols, styrene, titanium dioxide, and food-contact substances. EFSA.
Food and Agriculture Organization of the United Nations, & World Health Organization. (2024). Packaging and food safety: A holistic approach for human and environmental health. FAO.
Food Safety and Standards Authority of India. (2022–2024). Food Safety and Standards (Packaging) Regulation. FSSAI.
Franz, R., & Welle, F. (2022). Recycled plastics for food-contact applications: Safety evaluation and NIAS formation. Food Packaging and Shelf Life, 33, 100122. https://doi.org/10.1016/j.fpsl.2022.100122
Groh, K. J., Geueke, B., & Muncke, J. (2020). Food contact materials and chemicals: Current knowledge, challenges, and future needs. Environment International, 137, 105449. https://doi.org/10.1016/j.envint.2020.105449
Nerín, C., et al. (2023). Non-targeted screening strategies for non-intentionally added substances in food packaging using high-resolution mass spectrometry. Analytica Chimica Acta, 1267, 341424. https://doi.org/10.1016/j.aca.2023.341424
Thoden van Velzen, E. U., et al. (2023). Contaminants and non-intentionally added substances in mechanically recycled plastics: Challenges for a circular economy. Resources, Conservation & Recycling, 186, 106534. https://doi.org/10.1016/j.resconrec.2022.106534
U.S. Food and Drug Administration. (2023). Threshold of regulation (TOR) exemptions. FDA.
U.S. Food and Drug Administration. (2024). Food contact substances (FCS) notifications: Guidance and inventory. FDA.
